Pharma PES Capsules0 pages
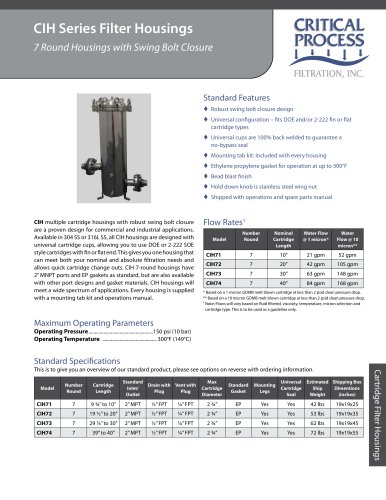
PPS Capsule Filters
Double Layered Polyethersulfone Membrane
Applications
Biologicals
Vaccines
WFI Water
LVPs and SVPs
Ophthalmics
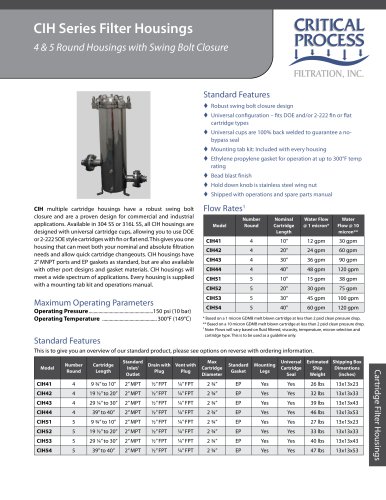
PPS Capsules are hydrophilic and manufactured with the highest
quality asymmetric polyethersulfone membrane, double layered
for extra security. Polyethersulfone (PES) membrane exhibits
excellent flow rates with high throughput. PPS capsules are
used for sterilizing filtration, the most critical applications in
the pharmaceutical industry. PPS capsule elements are 100%
integrity tested during production.
Validated for use in multiple pharmaceutical applications
Excellent flow rates with high throughput
Integrity testable
Designed for minimal leachables and extractables
Low adsorption of proteins and preservatives
Flow Rate / Filtration Area
The following table represents typical water flow at a one psi (69
mbar) pressure differential across a single 2 inch capsule with 1.0
ft² (930 cm²) of media with 1/2” FNPT ports. The test fluid is water
at ambient temperature. Higher pressure drops are acceptable,
but as flows increase the pressure drop of the housing becomes
more apparent.
Pore
Size
0.03
μm
0.10
μm
0.22
μm
0.45
μm
0.65
μm
0.8
μm
1.0
μm
1.2
μm
GPM
0.16
0.26
0.46
0.71
0.86
0.91
0.97
1.0
LPM
0.61
0.98
1.74
2.69
3.26
3.44
3.67
3.78
* For approximate flow rates for 5” through 30” capsules, refer to the
appropriate cartridge data sheet
Construction Materials
Housing
Polypropylene
Filtration Media
Double Layered Polyethersulfone (PES)
Membrane
Media Support
Polypropylene
End Caps
Polypropylene
Center Core
Polypropylene
Outer Support Cage
Polypropylene
Sealing Method
Thermal Bonding
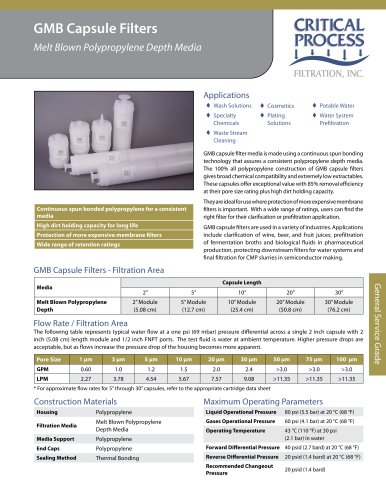
Specific applications for PPS capsule filters are final, sterilizing
filtration of USP Water for Injection (WFI), diagnostic solutions,
vaccines, ophthalmics, SVPs, LVPs and biological products.
Polyethersulfone is particularly suited for the filtration of products
that contain elements that can adsorb to the media, such as
preservatives and proteins. The lower binding characteristics
of polyethersulfone (PES) make it a good choice for filtration of
valuable protein solutions such as vaccines and biologicals as
well as ophthalmic solutions.
PPS Capsule Filters - Filtration Area
Media
PES
Membrane
Capsule Length
2”
5”
10”
20”
30”
1.0 ft2
(0.93m2)
3.0 ft2
(2.78m2)
7.0 ft2
(6.50m2)
14.0 ft2
(13.01m2)
21.0 ft2
(19.51m2)
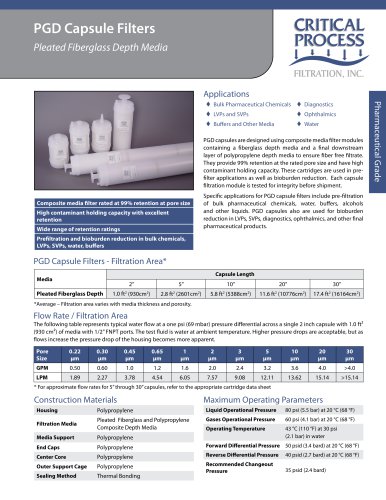
Sanitization/Sterilization
Autoclave........................... 250° F (121° C), 30 min, multiple cycles
Chemical Sanitization.......... Industry standard concentrations
of hydrogen peroxide, paracetic acid, sodium hypochlorite and
other selected chemicals.
Note................................PPS capsules are not to be used in steam.
Pre-Sterilized.............PPS capsules are offered in both non- and
pre-sterilized forms.
Maximum Operating Parameters
Liquid Operational Pressure
80 psi (5.5 bar) at 20 °C (68 °F)
Gases Operational Pressure
60 psi (4.1 bar) at 20 °C (68 °F)
Operating Temperature
43 °C (110 °F) at 30 psi
(2.1 bar) in water
Forward Differential Pressure 50 psid (3.4 bard) at 20 °C (68 °F)
Reverse Differential Pressure
40 psid (2.7 bard) at 20 °C (68 °F)
Recommended Changeout
Pressure
35 psid (2.4 bard)
Pharmaceutical Grade
Diagnostics
"
 عضویت
عضویت  ورود اعضا
ورود اعضا راهنمای خرید
راهنمای خرید