Drugs of Abuse Method Development Guide0 pages
D
CH3
N
N
D
H
O
C
O
N
D
BIOANALYSIS/DRUGS OF ABUSE
D
O
COOH
N
H
C
H
COO
H
C
H
O
O
CH 3
N
O
H
O
O
HC
C
H
OO
H
NH2
O
C
H
O
O
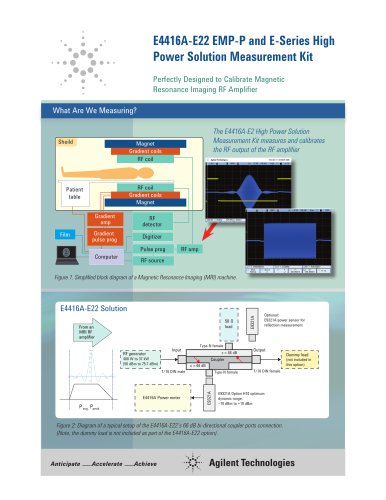
LC/MS methods are rapidly becoming an important
O
tool for drug screening and confirmation because
OO
they typically don’t require a derivatization step, and
are often less complicated than GC/MS methods. O
You can simplify and optimize method development
even further using new technologies from Agilent.
D
CD3O HC
H
H
C
N
CH 3
O
O
N
H
O
CH
H3 3
O
D
O
C
O
O
H
NH2
C
C
COOH D 3
D
H D D
NH
O
OD
H
CD3
D
COOH D
C
D H
C 3
H
COOH
H
H
NH 2
N
O
DD
O
H
H
C
C
D
OO C
N
D
D
D
D D
O
D
H
H
CH 3
D
N
N
D
D
OH
CO
D
C
O
H
O
H
OC
CD 3 CD 3
CH3
O
QUICK-REFERENCE METHOD GUIDE
N
C
H
N
H
CH 3
D
D
H
D
D
O
O
N
CH3
H D
N
D
H
N
COOH
N
H
D
N
D 3
CD
N
H
COO
C
DD H 3
D
N
D
COOH
N
D
D
CD 3
C
C
H
CD 3
C
C
NH 2
C
H
NH 2
D
D
O
H D
D
O
CH 3
KeyC workflow considerations:
H
NH 2
H2
H
NNH2
C C
C
C
•t epending on the C
analytical instruments used, sample matrices – such as
O
CD
H
H
Hblood, plasma, and urine – can adversely affect your system and columns.
H
H
H
Accordingly, reliable sample prep is an essential part of the workflow.
O
O
H3
•t olumns and sample prep techniques should beCselected with an eye
C
H
O
NH
toward return on investment – specifically, time and cost2 when compared
C C
to workflow performance and results.
O
H
H
•t hen using an electrospray source, ion suppression caused by sample
W
matrices can negatively impact your MS results.
O
•t any drugs of abuse are challenging to analyze because of factors, such as
M
volatility, hydrophobicity, and “stickiness” to glass and plastic.
D
CD3
C
C
O
D
H
O
NH2
"
 عضویت
عضویت  ورود اعضا
ورود اعضا راهنمای خرید
راهنمای خرید